- AdAlta’s lead drug for treatment of Idiopathic Pulmonary (lung) Fibrosis AD-114 redesigned as new molecule, called AD-214
- AD-214 retains unique therapeutic benefits of AD-114, but with greater potency and significantly improved half-life
- New design combines the i-body with an Fc fragment that enables the drug to stay in the body for longer, benefitting patients
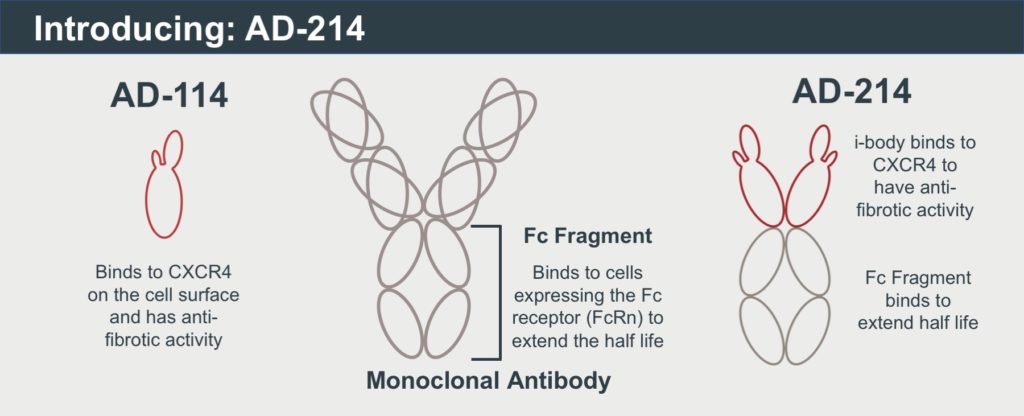
Australian biotech AdAlta (ASX: 1AD) today announced major improvements to the design of its preclinical i-body therapy for lung fibrosis, AD-114, which has significantly enhanced the drug’s potency and extended its half-life.
AdAlta has called its improved AD-114 molecule AD-214.
AD-214 contains two AD-114 i-body molecules at its front end, to increase its ability to bind to its human target, CXCR4, where it exerts its therapeutic effect. The addition of the Fc fragment (the tail region of a traditional monoclonal antibody) extends AD-214’s half-life, or duration of time in which it will stay in the body.
AdAlta CEO Sam Cobb said the Company will progress AD-214 into a Phase I clinical trial in the second half of 2019. Accordingly, the Company will prioritize development of AD-214 and halt further expenditure on manufacturing the current AD-114 molecule.
“AD-214 will ultimately be of greater benefit to future patients and potential commercial partners,” Cobb said.
“An increased half-life results in significantly less frequent dosing and therefore a more desirable and acceptable outcome for patients leading to greater drug compliance. Less frequent dosing is considered important for patients and their physicians when treating fibrotic diseases.
“AD-214 will widen the commercial interest when we progress our partnering discussions. Although the redesign of AD-114 into the new Fc-Fusion molecule AD-214 delays us from entering human clinical trials by around 12 months, AdAlta believes the timeline associated with this new molecule is more than offset by its broader clinical utility in a wider range of fibrotic diseases outside of Idiopathic Pulmonary Fibrosis”.
Dr Robert Peach, AdAlta’s Non-Executive Director who has a long history in the development of commercially available biologic drugs, including the Fc-Fusion drug blockbuster Orencia commented, “The data on AdAlta’s CXCR4 i-body antagonist is extremely encouraging for the treatment of multiple fibrotic diseases, including NASH, and age related macular degeneration.
“We have been able to leverage all the work done to date on AD-114 in the development of AD-214. This means we can make use of our existing strong data package and retain orphan drug status for treatment of idiopathic pulmonary fibrosis. We expect AD-214 will deliver increased commercial and therapeutic benefits in multiple fibrotic conditions, combined with the strong commercial precedent of existing mainstream drugs developed using the Fc-Fusion route reinforces the validity of this approach.”
There are greater than 70 antibodies currently on the market and eleven FDA approved Fc-Fusion proteins. There is a straightforward method of manufacturing and consequently it is expected that costs of goods can be reduced for AD-214 due to less frequent dosing and a well understood manufacturing pathway.
Learn more about AD-214 here, check out AdAlta’s latest company presentation and listen to an interview with CEO Sam Cobb.
AdAlta will also be holding Shareholder Briefing Sessions in Melbourne, Brisbane, Perth and Sydney in April, find out more information here.
